Tildipirosin CAS NO 328898-40-4 Inquire about Tildipirosin
Tecoland supplies Tildipirosin bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Tildipirosin is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Tildipirosin 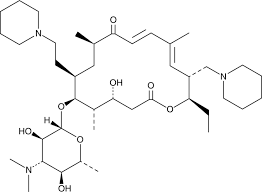
Tildipirosin is a semisynthetic derivative of the naturally occurring 16-membered macrolide tylosin. Tildipirosin is intended for parenteral treatment of respiratory disease in cattle and swine. Tildipirosin will be administered as a single-dose injection: subcutaneously in cattle and intramuscularly in swine. The anticipated optimal clinical dose is 4 mg/kg bw. Tildipirosin is not used in human medicine. It is marketed under the brand name Zuprevo. As for other macrolides, the antimicrobial activity of tildipirosin is due to its binding to the ribosomal 50S subunit of bacterial cells thereby inhibiting bacterial protein synthesis. The in vitro antimicrobial activity against Gram-negative and Gram-positive pathogens indicates that tildipirosin is effective against a range of bacterial pathogens frequently associated with bovine and swine respiratory disease. Comparison of minimum inhibitory versus bactericidal concentrations shows that generally the antimicrobial action of tildipirosin is bacteriostatic.
Side Effects
Tildipirosin may cause sensitization by skin contact.
Tilmicosin may be cardiotoxic in some animals. Injections to pigs have been fatal because of cardiotoxicity. The cardiac effects are increased heart rate and decreased contractility. However, administration of tilmicosin oral premix in feed of pigs has been safe. In dogs, tilmicosin injections have caused cardiac toxicosis and may be caused by calcium-channel blockade; it was reversed by administration of calcium. In goats, injections greater than 10 mg/kg IM or SQ can cause toxicity. In horses, injections of tilmicosin IM or SQ greater than 10 mg/kg can lead to toxicity.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.