Radezolid CAS NO 869884-78-6 Inquire about Radezolid
Tecoland supplies Radezolid bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Radezolid is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Radezolid?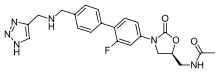
Radezolid (codenamed RX-1741) is a novel oxazolidinone antibiotic being developed by Melinta Therapeutics, Inc. It is used for the treatment of serious multi-drug–resistant infections. Radezolid has excellent in vitro and in vivo activity against a variety of Gram-positive bacteria including methicillin-resistant Staphylococcus aureus (MRSA). Effcacy has been attributed to the finding that radezolid accumulates in vitro in macrophages, polymorphonuclear leukocytes (PMNs), epithelial and endothelial cells.
Radezolid has completed two phase-II clinical trials. One of these clinical trials was for uncomplicated skin and skin-structure infections (uSSSI), and the other clinical trial was for community acquired pneumonia (CAP).
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.