Ibrutinib CAS NO 936563-96-1 Inquire about Ibrutinib
Tecoland supplies Ibrutinib bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Ibrutinib is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Ibrutinib 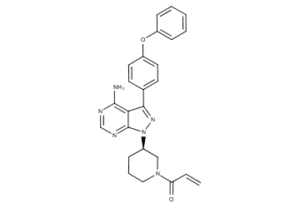
Ibrutinib (Imbruvica) is a type of medication called a kinase inhibitor, which is used to treat various blood cancers and also a serious complication of allogeneic stem cell transplants called chronic graft versus host disease (cGVHD).
Ibrutinib helps to slow down how quickly certain blood cancers progress by working against cancerous B cells, a type of white blood cell. It does this by blocking Bruton’s tyrosine kinase (BTK) signaling. BTK is a protein found on B cells that instructs B cells to remain alive and multiply. Ibrutinib also blocks the activity of a similar protein called interleukin-2-inducible T-cell kinase (ITK). It’s thought that this action and it’s ability to block BTK helps in cGVHD.
What is ibrutinib used for?
Ibrutinib is a prescription medicine used to treat :
- Adults with mantle cell lymphoma (MCL) who have received at least one prior treatment.
- Adults with chronic lymphocytic leukemia (CLL)/Small lymphocytic lymphoma (SLL).
- Adults with chronic lymphocytic leukemia (CLL)/Small lymphocytic lymphoma (SLL) with 17p deletion.
- Adults with Waldenström’s macroglobulinemia (WM).
- Adults with marginal zone lymphoma (MZL) who require a medicine by mouth or injection (systemic therapy) and have received a certain type of prior treatment.
- Adults and children 1 year of age and older with chronic graft versus host disease (cGVHD) after failure of 1 or more lines of systemic therapy.
It is not known if it is safe and effective in children under 1 year of age.
What should I tell my doctor before taking ibrutinib?
Before taking ibrutinib, tell your healthcare provider about all of your medical conditions, including if you:
- have had recent surgery or plan to have surgery. Your healthcare provider may stop ibrutinib for any planned medical, surgical, or dental procedure.
- have bleeding problems
- have or had heart rhythm problems, smoke, or have a medical condition that increases your risk of heart disease, such as high blood pressure, high cholesterol, or diabetes
- have an infection
- have liver problems
Side Effects of ibrutinib?
Ibrutinib may cause serious side effects, including:
- Bleeding problems (hemorrhage) are common during treatment with ibrutinib, and can also be serious and may lead to death. Your risk of bleeding may increase if you are also taking a blood thinner medicine. Tell your healthcare provider if you have any signs of bleeding, including:blood in your stools or black stools (looks like tar)- pink or brown urine- unexpected bleeding, or bleeding that is severe or that you cannot control
– vomit blood or vomit looks like coffee grounds
– cough up blood or blood clots
– increased bruising
– dizziness
– weakness
– confusion
– change in your speech
– headache that lasts a long time or severe headache
- Infections can happen during treatment with ibrutinib. These infections can be serious and may lead to death. Tell your healthcare provider right away if you have fever, chills, weakness, confusion, or other signs or symptoms of an infection during treatment with ibrutinib.
- Heart problems. Serious heart rhythm problems (ventricular arrhythmias, atrial fibrillation and atrial flutter), heart failure and death have happened in people treated with ibrutinib, especially in people who have an increased risk for heart disease, have an infection, or who have had heart rhythm problems in the past. Tell your healthcare provider if you get any symptoms of heart problems, such as feeling as if your heart is beating fast and irregular, lightheadedness, dizziness, shortness of breath, swelling of the feet, ankles or legs, chest discomfort, or you faint. If you develop any of these symptoms, your healthcare provider may do a test to check your heart (ECG) and may change your ibrutinib dose.
- High blood pressure (hypertension). New or worsening high blood pressure has happened in people treated with ibrutinib. Your healthcare provider may start you on blood pressure medicine or change current medicines to treat your blood pressure.
- Decrease in blood cell counts. Decreased blood counts (white blood cells, platelets, and red blood cells) are common with ibrutinib, but can also be severe. Your healthcare provider should do monthly blood tests to check your blood counts.
- Second primary cancers. New cancers have happened during treatment with ibrutinib, including cancers of the skin or other organs.Tumor lysis syndrome (TLS). TLS is caused by the fast breakdown of cancer cells. TLS can cause kidney failure and the need for dialysis treatment, abnormal heart rhythm, seizure, and sometimes death. Your healthcare provider may do blood tests to check you for TLS.
The most common side effects of ibrutinib in adults with B-cell malignancies (MCL, CLL/SLL, WM and MZL) include:
- diarrhea
- tiredness
- muscle and bone pain
- rash
- bruising
The most common side effects of ibrutinib in adults or children 1 year of age and older with cGVHD include:
- tiredness
- muscle and joint pain
- nausea
- low red blood cell count (anemia)
- fever
- stomach pain
- bruising
- muscle spasms
- pneumonia
- diarrhea
- mouth sores (stomatitis)
- headache
- low platelet count
- bleeding
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.