Loxoprofen CAS NO 68767-14-6 Inquire about Loxoprofen
Tecoland supplies Loxoprofen bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Loxoprofen is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Loxoprofen?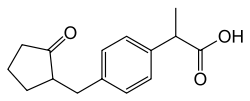
Loxoprofen is a nonsteroidal anti-inflammatory drug (NSAID) in the propionic acid derivatives group, which also includes ibuprofen and naproxen among others. It is available in some countries for oral administration. A transdermal preparation was approved for sale in Japan on January 2006.It was patented in 1977 and approved for medical use in 1986.
Mechanism of Action:
As most NSAIDs, loxoprofen is a non-selective cyclooxygenase inhibitor, and works by reducing the synthesis of prostaglandins from arachidonic acid.
Pharmacokinetics:
Loxoprofen is a prodrug. It is quickly converted to its active trans-alcohol metabolite following oral administration, and reaches its peak plasma concentration within 30 to 50 minutes.
Pharmacodynamics:
Loxoprofen is responsible for the formation of various biologically active pain, fever, and inflammatory mediators. These include prostaglandins, prostacyclin, thromboxane, and arachidonic acid.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.