Lapatinib Ditosylate CAS NO 388082-78-8 Inquire about Lapatinib Ditosylate
Tecoland supplies Lapatinib Ditosylate bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Lapatinib Ditosylate is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Lapatinib Ditosylate?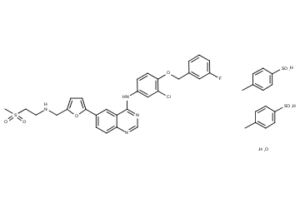
Lapatinib (INN), used in the form of lapatinib ditosylate, (USAN) (Tykerb/Tyverb, GSK) is an orally active drug for breast cancer and other solid tumours. It is a dual tyrosine kinase inhibitor which interrupts the HER2/neu and epidermal growth factor receptor (EGFR) pathways. It is used in combination therapy for HER2-positive breast cancer. It is used for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 (ErbB2).
What is the mechanism of action?
Lapatinib inhibits the tyrosine kinase activity associated with two oncogenes, EGFR (epidermal growth factor receptor) and HER2/neu (Human EGFR type 2). Over expression of HER2/neu can be responsible for certain types of high-risk breast cancers in women. Like Sorafenib, lapatinib is a protein kinase inhibitor shown to decrease tumor-causing breast cancer stem cells. Lapatinib inhibits receptor signal processes by binding to the ATP-binding pocket of the EGFR/HER2 protein kinase domain, preventing self-phosphorylation and subsequent activation of the signal mechanism (see Receptor tyrosine kinase#Signal transduction).
How to dose Lapatinib Ditosylate?
The recommended dose of lapatinib ditosylate is 1250 milligrams (mg) orally once daily on days 1 through 21 continuously in combination with capecitabine 2000 mg/squared meter/day orally divided into 2 doses (approximately 12 hours apart) on days 1 to 14, and repeated every 21 days. Lapatinib ditosylate doses should be taken at least one hour before or one hour after a meal. Capecitabine doses should be taken with food or within 30 minutes after food.
What are the side effects?
- Diarrhoea- This may occur in approximately 65% of patients (compared to 40% in patients treated with capecitabine alone) and is the most common reason for stopping treatment. If diarrhoea is severe it can lead to dehydration so aggressive treatment with anti-diarrhoea drugs and fluid and electrolyte replacement may be required.
- Nausea and vomiting- Affecting between 20-40% of patients receiving combination therapy.
- Rash- This occurs in approximately 28 percent of patients and approximately twice as common compared to capecitabine alone.
- Hand-foot syndrome- This is an unusual side effect causing numbness, tingling, redness, swelling and discomfort of hands and feet. It occurs in around 50% of patients.
- Fatigue and tiredness.
- Mouth sores.
- Loss of appetite.
Storage
Store at or below -20 ºC.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.