Everolimus CAS NO 159351-69-6 Inquire about Everolimus
Tecoland supplies Everolimus bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Everolimus is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Everolimus used for?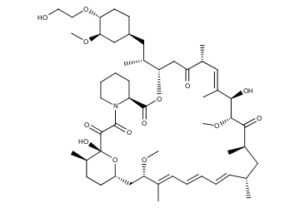
Everolimus is a new mTOR inhibitor drug used as an immunosuppressant to prevent rejection of organ transplants. It is related to Sirolimus (Rapamycin), and works in the same way.
Everolimus may have a role in heart transplantation as it has been shown to reduce chronic allograft vasculopathy in such transplants. It also may have a similar role to sirolimus in kidney and other transplants.
Researchers also reported that Everolimus significantly reduced the incidence of cytomegalovirus (CMV) infection, a major infection after transplantation and an important risk factor for the development of cardiac allograft vasculopathy and other problems.
Everolimus-FDA Approval Status
On August 30, 2004, Novartis Pharma AG received an “approvable” letter from the U.S. Food and Drug Administration (FDA) for everolimus in combination with cyclosporine, USP MODIFIED, for the prevention of rejection episodes following heart or kidney transplantation.
The FDA has requested that Novartis provide “additional” information supporting a safe and effective dosing regimen for the combination of everolimus and cyclosporine. The FDA had previously issued an approvable letter for Certican in October 2003 with a request for additional clinical data on dosing regimens. Novartis submitted data in response to the first approvable letter in February 2004. However, the FDA still believes that additional clinical studies on dosing are necessary.
Everolimus-A Proliferation Inhibitor
Everolimus appears to target many of the underlying causes of cardiac allograft vasculopathy (chronic rejection) in transplantation. The drug was developed by Novartis and has been submitted for regulatory review for use in both kidney and heart transplantation in the USA, Canada and the European Union.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.