Erlotinib HCl CAS NO 183319-69-9 Inquire about Erlotinib HCl
Tecoland supplies Erlotinib HCl bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Erlotinib HCl is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Erlotinib HCl?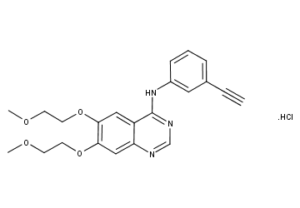
Erlotinib hydrochloride (trade name Tarceva) is a drug used to treat non-small cell lung cancer, pancreatic cancer and several other types of cancer. It is a reversible tyrosine kinase inhibitor, which acts on the epidermal growth factor receptor (EGFR). It is marketed in the United States by Genentech and OSI Pharmaceuticals and elsewhere by Roche. In lung cancer, it extends life by an average of 3.3 months at a cost of $95,000.
What is the mechanism of action?
Competitively inhibits binding at the ATP binding site with EGFR (HER1) tyrosine kinase.
How to dose Erlotinib HCl?
The recommended daily dose of TARCEVA for NSCLC is 150 mg taken on an empty stomach at least one hour before or two hours after the ingestion of food.
What is the side effects?
Common side effects include:Rash occurs in the majority of patients. This resembles acne and primarily involves the face and neck. It is self-limited and resolves in the majority of cases, even with continued use. Interestingly, some clinical studies have indicated a correlation between the severity of the skin reactions and increased survival though this has not been quantitatively assessed. The Journal of Clinical Oncology reported in 2004 that “cutaneous [skin] rash seems to be a surrogate marker of clinical benefit, but this finding should be confirmed in ongoing and future studies.” The newsletter Lung Cancer Frontiers reported in its October 2003 issue, “Patients with moderate to severe cutaneous reactions [rashes] have a far better survival, than those with only mild reactions and much better than those with no cutaneous manifestations of drug effects.”
Storage
Store at -20° C.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.