Doxorubicin HCl CAS NO 25316-40-9 Inquire about Doxorubicin HCl
Tecoland supplies Doxorubicin HCl bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Doxorubicin HCl is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Doxorubicin HCl?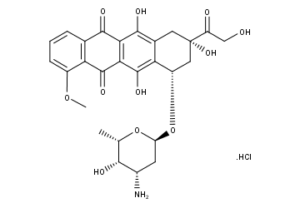
Doxorubicin HCl is a drug used in cancer chemotherapy. It is an anthracycline antibiotic, closely related to the natural product daunomycin, and like all anthracyclines it intercalates DNA. It is commonly used in the treatment of a wide range of cancers, including hematological malignancies, many types of carcinoma, and soft tissue sarcomas.
The drug is administered in the form of hydrochloride salt intravenously. It may be sold under the brand names Adriamycin PFS, Adriamycin RDF, or Rubex. It is photosensitive and it is often covered by an aluminum bag to prevent light from affecting it.
What is Doxorubicin used for?
Cancers treated with Doxorubicin include: bladder, breast, head and neck, leukemia (some types), liver, lung, lymphomas, mesothelioma, multiple myeloma, neuroblastoma, ovary, pancreas, prostate, sarcomas, stomach, testis (germ cell), thyroid, uterus.
How is Doxorubicin administered?
Doxorubicin is given through a vein by intravenous injection (IV). The syringe needle is placed directly into the vein or central line and the drug is given over several minutes. Doxorubicin can also be given by continuous infusion. Rarely, Doxorubicin is given by injection into an artery. There is no pill form of Doxorubicin.
Doxorubicin is a vesicant. A vesicant is a chemical that causes extensive tissue damage and blistering if it escapes from the vein. The or doctor who gives Doxorubicin must be carefully trained. If you notice redness or swelling at the IV site while you are receiving Doxorubicin, alert your health care professional immediately.
The amount of Doxorubicin you will receive depends on many factors, including your height and weight, your general health or other health problems, and the type of cancer you have. Your doctor will determine your exact dosage and schedule.
Side Effects of Doxorubicin:
Important things to remember about the side effects of Doxorubicin: You will not get all of the side effects mentioned below.
- Side effects are often predictable in terms of their onset, duration, and severity.
- Side effects are almost always reversible and will go away after therapy is complete.
The following side effects are common (occurring in greater than 30%) for patients taking Doxorubicin:
Early Side Effects: (within one week after treatment begins)
- Pain along the site where the medication was given.
- Nausea or vomiting
Later Side Effects: (within two weeks after treatment begins)
- Low blood counts.Your white and red blood cells and platelets may temporarily decrease.This can put you at increased risk for infection, anemia and/or bleeding.
- Mouth sores
- Hair loss on the scalp or elsewhere on the body (called alopecia). Most patients do lose some or all of their hair during their treatment. But your hair will grow back after treatment is completed.
The following side effects are less common (occurring in 10-29%) for patients taking Doxorubicin:
Early: (within one week after treatment begins)
- Eyes watering
- Urine may appear red, red-brown, orange or pink from the color of the medication for one to two days after you receive a dose.
Later: (within two weeks after treatment begins)
- Darkening of the nail beds.
- Darkening of the skin where previous radiation treatment has been given.
Problems with fertility ability to bear children. (occurs in about 10% of both men and women this should be discussed with your doctor prior to therapy).
A serious but uncommon side effect of Doxorubicin can be interference with the pumping action of the heart. You can receive only up to a certain amount of Doxorubicin during your lifetime. This lifetime maximum dose may be lower if you have heart disease risk factors such as radiation to the chest, advancing age, and use of other heart-toxic drugs. Your doctor will check your heart function before you may take any Doxorubicin and will monitor your heart closely during your treatment. Dose-related heart problems can occur as late as 7 or 8 years after treatments have ended.
The following symptoms require medical attention, but are not emergency situations. Contact your health care provider within 24 hours of noticing any of the following:
- Mouth sores (painful redness, swelling or ulcers)
- Nausea (interferes with ability to eat and unrelieved with prescribed medication)
- Vomiting (vomiting more than 4-5 times in a 24 hour period)
- Diarrhea (4-6 episodes in a 24-hour period)
- Fast or irregular heart beats
- Unusual bleeding or bruising
- Black or tarry stools, or blood in your stools or urine
- Extreme fatigue (unable to carry on self-care activities)
- Swelling of the feet or ankles
- Doxorubicin Precautions
Before starting Doxorubicin treatment, make sure you tell your doctor about any other medications you are taking (including over-the-counter, vitamins, or herbal remedies). Do not take aspirin or products containing aspirin unless your doctor permits this.
Do not receive any kind of vaccination without your doctor approval while taking Doxorubicin.
For both men and women: Use contraceptives, and do not conceive a child (get pregnant) while taking Doxorubicin. Barrier methods of contraception, such as condoms, are recommended. Discuss with your doctor when you may safely become pregnant after therapy.
People with congestive heart failure, those who have already had high doses of Doxorubicin or a similar drug, and those with permanent problems with blood counts (bone marrow suppression) cannot receive Doxorubicin.
How does Doxorubicin Work?
Cancerous tumors are characterized by cell division, which is no longer controlled as it is in normal tissue. Normal cells stop dividing when they come into contact with like cells, a mechanism known as contact inhibition. Cancerous cells lose this ability. Cancer cells no longer have the normal checks and balances in place that control and limit cell division. The process of cell division, whether normal or cancerous cells, is through the cell cycle. The cell cycle goes from the resting phase, through active growing phases, and then to mitosis (division).
The ability of chemotherapy to kill cancer cells depends on its ability to halt cell division. Usually, the drugs work by damaging the RNA or DNA that tells the cell how to copy itself in division. If the cells are unable to divide, they die. The faster the cells are dividing, the more likely it is that chemotherapy will kill the cells, causing the tumor to shrink. They also induce cell suicide (self-death or apoptosis).
Chemotherapy drugs that affect cells only when they are dividing are called cell-cycle specific. Chemotherapy drugs that affect cells when they are at rest are called cell-cycle non-specific. The scheduling of chemotherapy is set based on the type of cells, rate at which they divide, and the time at which a given drug is likely to be effective. This is why chemotherapy is typically given in cycles.
Unfortunately, chemotherapy does not know the difference between the cancerous cells and the normal cells. Chemotherapy will kill all cells that are rapidly dividing. The normal cells will grow back and be healthy but in the meantime, side effects occur. The normal cells most commonly affected by chemotherapy are the blood cells, the cells in the mouth, stomach and bowel, and the hair follicles; resulting in low blood counts, mouth sores, nausea, diarrhea, and/or hair loss. Different drugs may affect different parts of the body.
Doxorubicin is classified as an antitumor antibiotic. Antitumor antibiotics are made from natural products produced by species of the soil fungus Streptomyces. These drugs act during multiple phases of the cell cycle and are considered cell-cycle specific. There are several types of antitumor antibiotics.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.