Dasatinib CAS NO 302962-49-8 Inquire about Dasatinib
Tecoland supplies Dasatinib bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Dasatinib is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Dasatinib?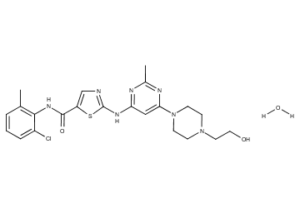
Dasatinib, previously known as BMS-354825, is a cancer drug produced by Bristol-Myers Squibb and sold under the trade name Sprycel. Dasatinib is an oral multi- BCR/ABL and Src family tyrosine kinase inhibitor approved for use in patients with chronic myelogenous leukemia (CML) after imatinib treatment and Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). It is being evaluated for use in numerous other cancers, including advanced prostate cancer.
What is the mechanism of action?
Dasatinib inhibits multiple tyrosine kinases including BCR-ABL, the fusion protein created by the abnormal Philadelphia chromosome (Ph) which characterizes chronic myeloid leukemia. 2,3 Competitive inhibition at the enzyme’s ATP-binding site leads to inhibition of tyrosine phosphorylation of proteins involved in BCR-ABL signal transduction. 2,3 Inhibition is not completely selective, as dasatinib also inhibits other kinases including the Src family (LYN, HCK), c-kit, ephrin receptor (EPH), and platelet-derived growth factor receptor (PDGF?). 2,3 Dasatinib is structurally unrelated to imatinib and is approximately 300-fold more potent in terms of BCR-ABL inhibition. 3,4 Dasatinib may overcome imatinib resistance that results from BCR-ABL kinase domain mutations, activation of alternate signaling pathways involving the Src family kinases, and multi-drug resistance gene over expression.
How to dose Dasatinib?
The recommended dose range of dasatinib is 100-180 mg orally once daily. For newly diagnosed Ph+ CML treatment starts at 100 mg and is increased to 140 mg daily as needed. For advanced Ph+ CML or Ph+ ALL treatment is started at 140 mg and increased to 180 mg daily as needed.
What is the side effect?
Common side effects include fluid retention, headache, diarrhea, constipation, weakness, nausea and vomiting, abdominal distention, weight loss or gain, rash, itching, chills, dizziness, and muscle pain. Serious side effects include fever associated with reduced white blood cells, reduced platelets, and red blood cell counts, infection, stomach or intestinal bleeding, bleeding in the brain, heart failure, and fluid in the lungs.
Storage
Dasatinib should be stored at room temperature between 15-30 C (59-86 F).
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.