Axitinib CAS NO 319460-85-0 Inquire about Axitinib
Tecoland supplies Axitinib bulk active pharmaceutical ingredient (API) to the pharmaceutical industry. Our Axitinib is manufactured by cGMP compliant facility. Welcome to contact us for further details including current DMF status for the product and up to date regulatory status of the manufacturing facility. We look forward to assisting you with your research and development projects.
What is Axitinib?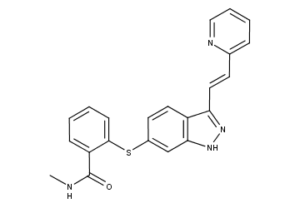
Axitinib (AG013736; trade name Inlyta) is a small molecule tyrosine kinase inhibitor developed by Pfizer. It inhibits multiple targets, including VEGFR-1, VEGFR-2, VEGFR-3, platelet derived growth factor receptor (PDGFR), and cKIT (CD117). It has been shown to significantly inhibit growth of breast cancer in xenograft models and has been successful in trials with renal cell carcinoma (RCC) and several other tumor types.
A Phase II clinical trial showed good response in combination chemotherapy with gemcitabine for advanced pancreatic cancer. However, Pfizer reported on January 30, 2009 that Phase III clinical trials of the drug when used in combination with gemcitabine showed no evidence of improved survival rates over treatments using gemcitabine alone for advanced pancreatic cancer and halted the trial.
In 2010, a Phase III trial for previously treated metastatic renal cell carcinoma (mRCC) showed significantly extended progression-free survival when compared to sorafenib.
In December 2011, the Oncologic Drugs Advisory Committee (ODAC) voted unanimously to recommend the approval of axitinib for the second-line treatment of patients with advanced renal cell carcinoma (RCC), based on the results of the Phase III trial comparing axitinib and sorafenib.
On January 27, 2012, the U.S. Food and Drug Administration (FDA) approved axitinib for use in patients with renal cell carcinoma that had failed to respond to a previous treatment.
Disclaimer:
Information on this page is provided for general information purposes. You should not make a clinical treatment decision based on information contained in this page without consulting other references including the package insert of the drug, textbooks and where relevant, expert opinion. We cannot be held responsible for any errors you make in administering drugs mentioned on this page, nor for use of any erroneous information contained on this page.